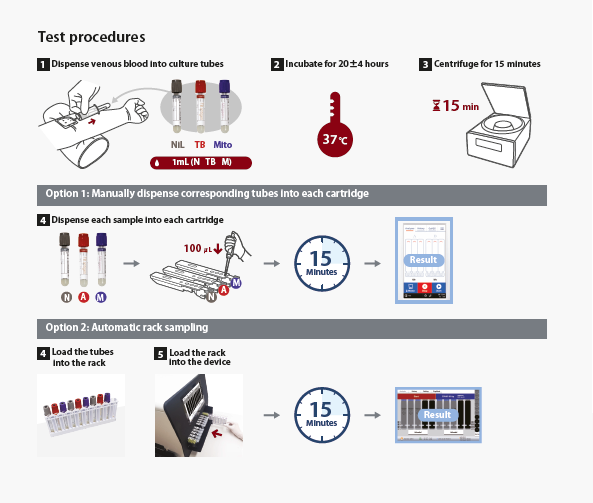
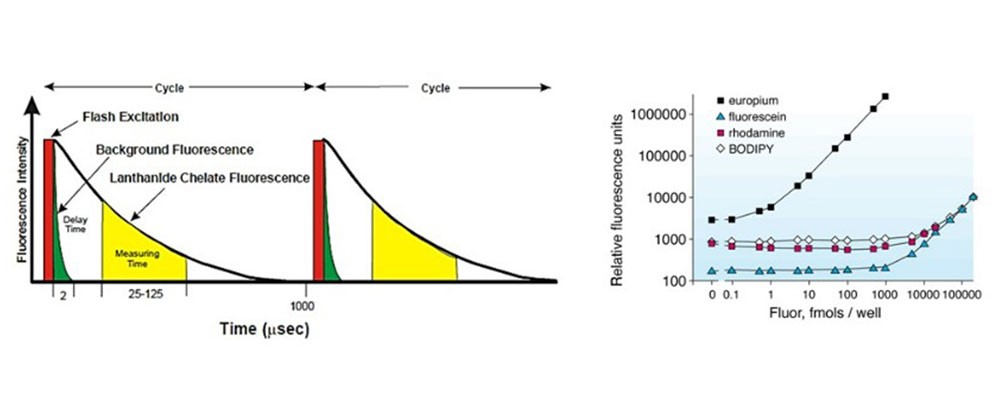
In the TRF method (Time Resolved Fluorescence), the light source does not emit continuous light radiation, but emits flashes of light (red rectangle). Since chromogen (europium chelates) has a certain inertia, it continues to emit fluorescence for a short period even after the flash of light (black line in the figure), as well as any background fluorescence (green area in the figure). The system detects only the residual fluorescence between one flash of light and the next (yellow area in the figure), where there is neither direct light from the source or that generated by the background radiation.
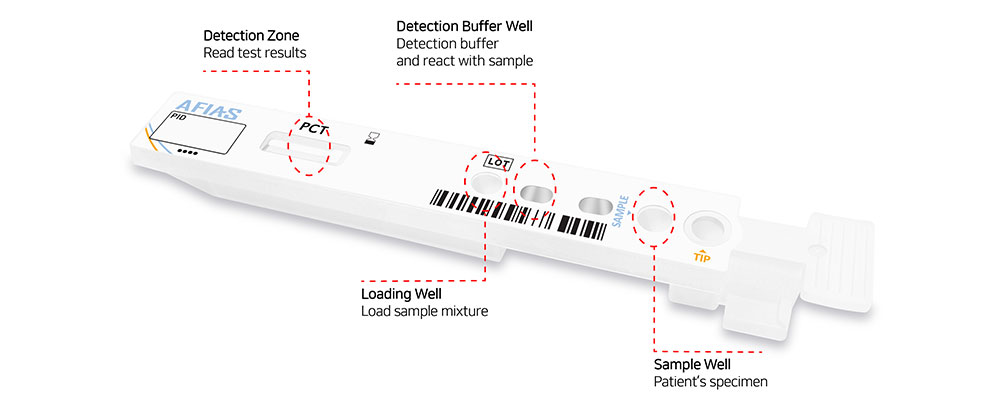
The innovative and disposable all-in-one cartridge is based on a lab-on-cartridge architecture: it contains all the necessary reagents to run the chemical reaction and enables a full automation of the testing.